Introduction and background
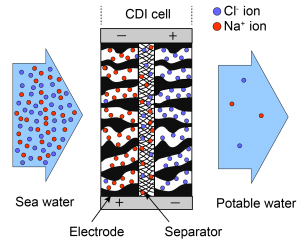 Capacitive deionization (CDI) is an emerging alternative technique for water desalination. It is especially promising for treating water with low and moderate salt concentration, also known as brackish water. The key component of a CDI cell is a pair of porous carbon electrodes. Salt ions are removed from water and held electrostatically at the pore surface. CDI operates at low voltage (<1.4V) and low pressure, and has the potential to be cost effective and energy efficient.
Capacitive deionization (CDI) is an emerging alternative technique for water desalination. It is especially promising for treating water with low and moderate salt concentration, also known as brackish water. The key component of a CDI cell is a pair of porous carbon electrodes. Salt ions are removed from water and held electrostatically at the pore surface. CDI operates at low voltage (<1.4V) and low pressure, and has the potential to be cost effective and energy efficient.
Traditional CDI cell designs rely on standard, low permeability porous carbon electrode materials and so use a “flow-between” architectures where the salt solution flows primarily through a space between two porous carbon electrodes. This traditional CDI architecture has charging times limited by the diffusion time associated with the space, increasing required package volume for a given flow rate and typically limiting salt reduction per stage to about 1 mM under normal operation.
The collaboration between Stanford Microfluidics Laboratory and Lawrence Livermore National Laboratory develops a new type of CDI cell which leverages novel, low-hydrodynamic resistance (high permeability) porous electrode materials and so uses a “flow-through” architecture wherein water is driven directly through the electrodes. We have shown that our flow-through CDI design can achieve 4-10 times faster charging times and up to 70 mM reduction in salt concentration. Our novel flow-through CDI architecture is ideally suited for compact, light-weight, and relatively fast-acting, solar-powered appliances.
MODELING A FLOW-THROUGH DEIONIZATION(FT-CDI) SYSTEM FOR ENHANCED SALT REMOVAL
I develop a one-dimensional modified Donnan (mD) ft-CDI model combining modified Nernst-Plank transport equation and macroscopic porous electrode theory. The model is able to predict desalination performance under various operation conditions. The simulation results demonstrate excellent agreement with experimental trends.
I use this model to predict effluent salt concentration of a ft-CDI cell under constant voltage (CV) or constant current (CC) operation conditions. The simulation results have revealed that under CC operation, the concentration profile inside the cell gradually develops to a quasi-steady state and produces constant effluent concentration. For CV operation, the cell demonstrates rapid desalination at the beginning, but the cell quickly reaches to saturation state after time constant.
This study expands understandings of ft-CDI cell performance and provides guidance for cell design and operation to improve total salt removal while maintaining energy efficiency.
Video 1 & Figure 1: Simulated salt concentration inside a ft-CDI cell under CV operation
Video 2 & Figure 2: Simulated salt concentration inside a ft-CDI cell under CC operation
(From let to right are left porous electrode, separator and right porous electrode in a cell assembly. Salt water flows from left to right. Unit: mM)
Characterization of resistances of a capacitive deionization system
For this project, I characterized electric resistances in a CDI system, with a focus on the resistance associated with the contact between current collectors and porous electrodes. I developed an equivalent circuit model to describe resistive components in a CDI cell. I proposed measurable figures of merit to characterize cell resistance. My work also shows 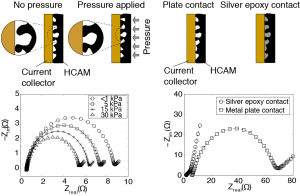 that contact pressure between porous electrodes and current collectors can significantly reduce contact resistance. Lastly, I proposed and tested an alternative electrical contact configuration which uses a pore-filling conductive adhesive (silver epoxy) and achieves significant reductions in contact resistance.
that contact pressure between porous electrodes and current collectors can significantly reduce contact resistance. Lastly, I proposed and tested an alternative electrical contact configuration which uses a pore-filling conductive adhesive (silver epoxy) and achieves significant reductions in contact resistance.

Nice one!